Today we’re going to take a look at an interesting prospect in the early stage Biotech world – CytRx Corporation (ticker: CYTR). CytRx is focused on oncology drugs and has three primary drug candidates in the mid-late stage pipeline phases with prospects for various indications.
Pipeline Review
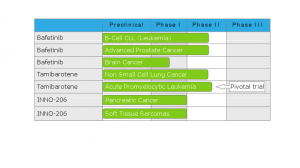
Here’s a recent snapshot of the pipeline indicating various drug candidates and their progression through the clinical phases required for eventual commercial distribution:
While this chart highlights multiple drug candidates and indications, I’m going to delve into Bafetinib in more detail.
Bafetinib
Bafetinib is currently at Phase II for B-CLL (refractory B-cell chronic lymphocytic leukemia) and prostate cancer. Additionally, there is a brain cancer indication at Phase I. The platform for this candidate is as a Tyrosine kinase inhibitor. Since the mechanism of action for this candidate is known, the clinical risk is likely lower than a novel mechanism based on other biotech candidates historically.
- Delivery – Bafetinib is delivered as an oral, which is a more desired route of administration than the common I.V. and injectable routes with many cancer drugs.
- Licensing – This novel candidate was initially developed by Nippon Shinayaku. As such, CytRx has an exclusive marketing agreement (ex-Japan) with Nippon Shinayaku whereby CytRx is obligated to pay up to $13.5 Million based on various US and EU milestones as well as royalties and sub-licensing income that isn’t publicly disclosed at this time.
- Clinical Trials for B-CLL – Phase I for B-CLL (refractory B-cell chronic lymphocytic leukemia) was completed in November 2008. Since some patients are intolerant of the popular drug Gleevec due to a particular gene mutation, it was encouraging to see many of these patients respond to Bafetinib. The trial was conducted at various sites and an optimal dose was determined at that stage. Following these outcomes, a Phase II proof of concept study was initiated in May 2010 as a second-line treatment for B-CLL. The trial targeted patients who had failed treatment with first-line treatments.
- B-CLL – Evidently, in the US alone, there are 16,000 new CLL cases per year and 4,400 deaths (Wikipedia).  Rates of CLL are elevated for people who were exposed to certain chemicals including Agent Orange.  The prognosis depends on the subtype, but across all types, the survival rate for all forms of CLL is only 5 years.
- Prostate Cancer and Brain Cancer Trials – Bafetinib also began a Phase II trial to evaluate the safety and efficacy of the drug for treatment of prostate cancer in September 2010. Additionally, in November 2010, a pharmacokinetic trial began to evaluate performance in brain tumor patients. In a recent analyst report from Zacks Research, the market for these various indications was pegged at over $1 Billion annually.
Disclosure
This is part of a paid, but independent Research Series on CytRx. The views and opinions expressed in this Series are purely my own. I have no positions in CytRx, and no plans to initiate any positions within the next 72 hours. Rule 17B requires disclosure of payment for investor relations services. www.darwinsmoney.com has been compensated $200 by a third party on 5/24/2011 for an independent review advertisement for CytRx. For complete disclosure/disclaimer statement, please review the Disclosure Statement relevant to this Series. [Link to Disclosure Policy].

{ 0 comments… add one now }
{ 1 trackback }